Video lecture for Atmospheres section
What temperature is and how it relates to a particle's speed and mass and how a gas' temperature affects the pressure has been discussed above but what will raise or lower the temperature of the air, particularly the air at the surface of a terrestrial world or the upper layers of a jovian planet's atmosphere? The surface temperature of a terrestrial planet is determined by how much energy the planet receives from the Sun and how quickly it radiates that solar energy back to space. As described in a later section, a terrestrial planet's interior temperature is determined by its size. The crust is a very poor conductor of any heat from the interior so the surface heat is all from the Sun. Jupiter, Saturn, and Neptune have extra heat energy coming from their interiors. Recall from the section about seasons that in order to keep the temperature the same, there must be a balance between the solar energy flowing onto the planet and the energy radiated back out to space.
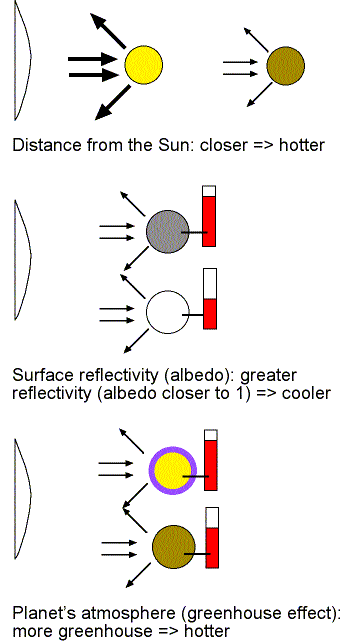
On global scales, three things can affect this energy flow and therefore, the average global surface temperature. As shown in the figure below, they are the planet's distance from the Sun, the planet's surface reflectivity (albedo), and the planet's atmosphere (through a process called the greenhouse effect).

Planets closer to the Sun receive more solar energy by an amount that depends on their distance squared (recall how light spreads out as described in the telescope chapter). Also recall from the light chapter that hotter dense things produce more energy (they are brighter). With more solar energy flowing to the closer planets, they must be hotter to re-radiate that energy back out to space. The amount of solar energy reflected immediately out to space is determined by the material on the planet's surface or clouds in the atmosphere. The fraction of sunlight that is reflected from an object is the albedo. If the albedo is closer to 1 (100% reflectivity), the planet does not need to be as hot to have its outflow of energy balance the inflow of solar energy. Darker objects absorb more solar energy and, therefore, they need to heat up more to re-radiate that energy back out to balance the inflow of solar energy. For example, you probably notice the difference between wearing a white T-shirt vs. wearing a black T-shirt outside on a sunny, summer day. Another example is the liquid water in our oceans absorb more solar energy than the ice areas at our poles.
Let's pause here and find out what the surface temperature should be for some of the planets. The rate of energy absorbed by the planet equals (the absorbing area of the planet) x (brightness of sunlight at the planet's distance from the Sun) x (fraction of sunlight absorbed). The rate of energy radiated by the planet using the discussion in the light chapter (see item 5 in the list on that webpage) equals (the surface area of the planet) x (energy radiated by each square meter every second, which changes as temperature to the fourth power). Setting the rate of solar energy absorbed equal to the rate of energy radiated by the planet, you find using the values for the distances and albedo given in the planet tables appendix, that Mercury should have an average temperature of 160 deg C, Venus should be -42 deg C, Earth should be -19 deg C, and Mars should be -63 deg C. Their actual temperatures are: Mercury = 425 deg C (day)/ -175 deg C (night) = midway value of 125 deg C; Venus = 464 deg C; Earth = 15 deg C; and Mars = -31 deg C (day)/-89 deg C (night) = midway value of -60 deg C. Mercury has such a large variation between day and night as well as a large variation between the daytime polar temperature of 317 deg C and daytime equatorial temperature of 452 deg C that the rather arbitrarily chosen midway value of 125 deg is close enough to the calculated value of 160 deg C.
How do you do that?To find the expected temperature of a planet, you need to set the rate of energy radiated by the planet equal to the rate of energy absorbed by the planet. Using the terms in the main text above, you find
where R is the radius of the planet, d is the distance from the Sun, a is the albedo, Lsun is the luminosity of the Sun (how much energy it emits every second), T is the temperature of the planet, and
Plugging in the values for the constants from the astronomical constants table in the appendix, you find
if the distance is measured in astronomical units. Finally, to convert to degrees Celsius, subtract 273 from the Kelvin value. For example, for the Earth, the albedo is 0.306 and the distance is 1.000 AU, so the expected temperature is 254 K or -19 C ---significantly below the freezing point of water! |
The difference between actual temperature values and the calculated values for the other three planets are a bit more interesting because of the effect of their atmospheres. A planet's atmosphere can hinder the rate that energy flows outward to space from the warm ground so the ground must heat up to increase the energy leaking out enough to balance the inflow of solar energy. This blanket effect of the atmosphere is called the greenhouse effect and is described in detail below. A planet's atmosphere can also hinder the rate that energy flows inward from space to the ground. The Moon and the Earth are at the same distance from the Sun but the Moon has a very large change in temperature from day to night due to the Moon's lack of an atmosphere. The Moon's surface temperature at its equator ranges from about 100 K (-173 deg C) at night to almost 400 K (127 deg C) during the day!

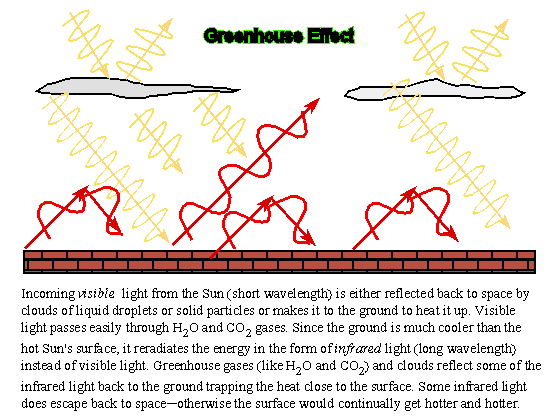
The greenhouse effect is named after the glass houses used to keep plants warm during cold weather. Energy in the form of visible light from the Sun passes through the glass walls and glass roofs of a greenhouse and heats up the plants and soil inside the greenhouse. The air in contact with the plants and soil gets warmed up. The glass walls and roofs prevent the hot air from escaping to the outside. The same sort of thing happens to the interior of your car when you leave it out in the Sun with the windows rolled up.
On a planet, certain gases like carbon dioxide or water vapor in the atmosphere prevent heat energy in the form of infrared light from leaking out to space. These so-called "greenhouse gases" allow visible light from the Sun to pass through and heat up the surface. A planet's surface is warm enough to emit infrared light. Some of the infrared light is absorbed by the greenhouse gases and radiated back toward the surface, warming the surface even more. Some of the energy is radiated back toward space. The surface warms enough so that the amount that does leak back out to space balances the solar energy flow inward. Note that if the greenhouse was a perfect blanket, then the surface would continue to get hotter and hotter.
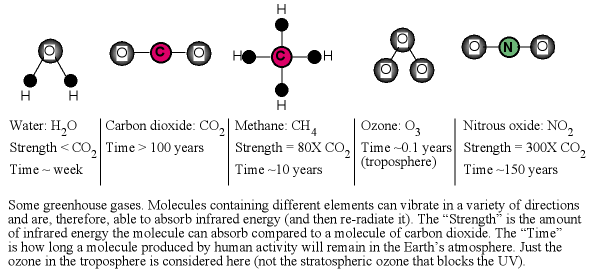
The primary greenhouse gases found in the atmospheres of our solar system's planets are given in the figure below. Not shown are chlorofluorocarbons (CFC's) and hydrofluorocarbons (HFC's) that are synthesized by humans. On the Earth the relative amounts these molecules contribute to the total greenhouse effect occurring are approximately: 60% for water, 26% for carbon dioxide, 5% for methane, 4% for ozone, 4% for the CFC's/HFC's, and 2% for nitrous oxide (rounding of the numbers to integer values means they will not add up to exactly 100%).
Now back to our three planets. Venus' actual temperature is over three times more than if there was no greenhouse effect at work. Earth has a natural greenhouse effect mostly caused by water vapor to raise the temperature by about 34 deg C so the oceans do not freeze. Mars has only a very slight warming because of its thin atmosphere.
Planet atmospheres have a layered structure based on how the temperature changes with increasing altitude. The greenhouse effect plays a major role for the lowest layer of a terrestrial planet's atmosphere and other heating agents can raise the temperature of the upper layers. In this section we will take a look at the Earth's atmosphere layers, then compare it to the other terrestrial planet atmospheres, and finally finish with the structures of the jovian planets atmospheres.
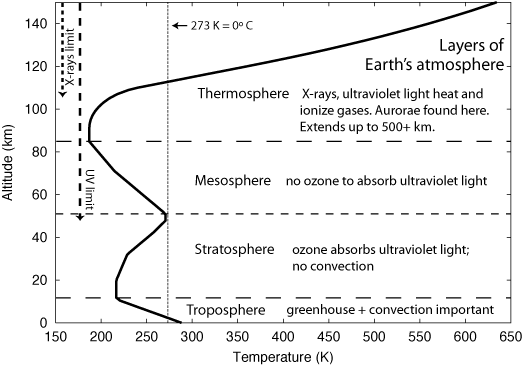
The figure above shows the bottom four layers of the Earth's atmosphere. (Standard model atmosphere from Steven Pietrobon at Small World Communications.) Here are short descriptions of each layer.
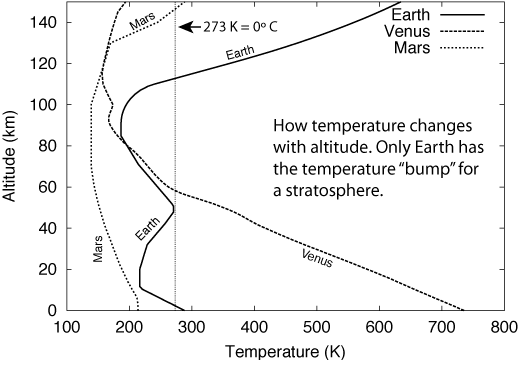
Both Mars and Venus have tropospheres of greater extent than the Earth, though for different reasons. Mars' atmosphere is much thinner than Earth's and there is less compression because of Mars' weaker gravity. Although Venus has weaker gravity than Earth, it has over ninety times the amount of atmosphere because of a runaway greenhouse effect that occurred at least hundreds of millions of years ago. That will be described further later but it does provide a warning to us that drastic global climate change is possible. Because their tropospheres extend over a greater distance than the Earth's troposphere, Mars' and Venus' clouds are found at higher altitudes. Mars and Venus also have thermospheres. What is missing is the temperature bump of a stratosphere (and mesophere) because they do not have an ozone layer to absorb the ultraviolet light. (Planet atmosphere models courtesy of Jere Justus at the Marshall Space Flight Center, NASA.)
The jovian planets have the same atmosphere layers as the Earth, though, their compositions, of course, are very different. Jupiter's atmosphere structure is described in the figure below.
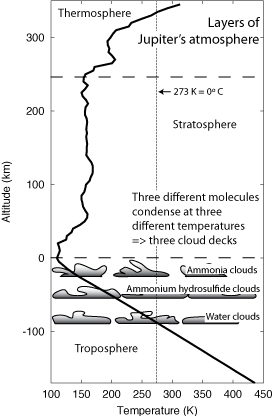
Jupiter's troposphere extends much further down merging smoothly into its interior. Mixed in with the abundant molecular hydrogen and helium are trace amounts of ammonia, water, and methane. Even smaller amounts of hydrogen sulfide (the stinky stuff of rotten eggs), other hydrogen polysulfides, and phosphorus are also present. Ammonia and hydrogen sulfide will mix together in water to make ammonium sulfide. Molecules of ammonia, ammonium hydrosulfide, and water will form droplets (condense) when the temperature is low enough. They will condense (and freeze) at different temperatures, though, so clouds of these molecules will form at different depths in the troposphere. There are three main clouds decks on Jupiter. Water condenses at a higher temperature than the other two, so water clouds are thought to exist at the deepest cloud layer. Higher up the temperature is low enough for the ammonium hydrosulfide to condense. Finally, just below the upper bound of the troposphere, the temperature is cold enough for ammonia to condense. Note that the cloud layers mark the upper bound of that type of molecule in the hydrogen/helium atmosphere.
The Galileo spacecraft dropped a probe into Jupiter's atmosphere when it arrived at Jupiter in December 1995. The probe got down to a depth of 161 km below the cloud tops before the probe stopped functioning because of high pressures and temperatures. At that depth the pressure was 22 bars and the temperature about 425 K. From the atmosphere structure figure above, the Galileo probe should have penetrated to where the water vapor is but it did not find the water. Unfortunately, the probe entered one of the clear, dry areas produced by downdrafts. Although, the probe got over 160 km below the cloud tops, it was still in the troposphere and its deepest point represents just 0.3% of Jupiter's radius. Above the troposphere of Jupiter, ultraviolet heating makes a stratosphere but other molecules instead of ozone absorb the ultraviolet light. At the highest levels are the thermosphere and the exosphere.
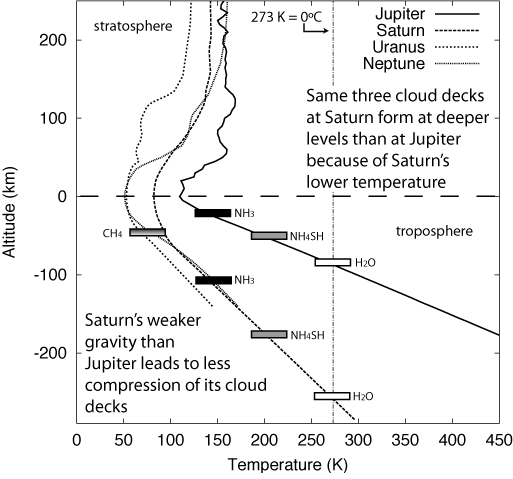
Saturn has the same three main cloud decks, though they are found at lower altitudes than on Jupiter because of Saturn's lower temperature (it is further from the Sun). Also, Saturn's cloud decks are further apart from each other because of Saturn's lower gravity---there is less compression of the gases. Because Saturn's clouds form at deeper positions in its troposphere, its cloud patterns appear more muted than on Jupiter. It is thought that the three cloud decks (of ammonia, ammonium hydrosulfide, and water) would be found much too deep in the tropospheres of Uranus and Neptune for us to see. Instead, Uranus and Neptune being even further from the Sun have cold enough tropospheres for methane to condense and freeze to form clouds (Jupiter and Saturn are too warm for methane clouds). Neptune has extra heat energy from its interior so its atmosphere temperature is warmer than Uranus.
In the next section we will take a look at how the gases move about and distribute energy.
![]() Go back to previous section --
Go back to previous section --
![]() Go to next section
Go to next section
last updated: October 28, 2024